32 The Plasma Membrane
Cells closely control the exchange of substances in and out of the cell. Some substances are excluded, others are taken in, and still others are excreted – all in controlled quantities. Although the plasma membrane encloses the cell’s borders, it is far from being a static barrier; it is dynamic and constantly in flux. The plasma membrane must be sufficiently flexible to allow certain cells, such as red blood cells and white blood cells, to change shape as they pass through narrow capillaries. In addition to these more obvious functions, the surface of the plasma membrane carries markers which allow cells to recognize one another. This is vital as these markers play a role in the “self” versus “non-self” distinction of the immune response.
The plasma membrane also carries receptors, which are attachment sites for substances that interact with the cell. Each receptor is structured to bind with a specific substance. The binding of a specific substance to its receptor on the plasma membrane can activate processes within the interior of the cell – such as activating enzymes involved in metabolic pathways. These metabolic pathways might be vital for providing the cell with energy, making substances for the cell, or breaking down cellular waste or toxins for disposal. Likewise, extracellular hormones and neurotransmitters bind to plasma membrane receptors which transmit a signal into the cell to intracellular molecules. Some recognition sites are used by viruses as attachment points. Although they are highly specific, pathogens like viruses may evolve to exploit receptors to gain entry to a cell by mimicking the specific substance that the receptor is meant to bind. This specificity helps to explain why human immunodeficiency virus (HIV) or any of the five types of hepatitis viruses invade only specific cells.
Fluid Mosaic Model
In 1972, S. J. Singer and Garth L. Nicolson proposed a new model of the plasma membrane. This theory, compared to earlier theories, best explains both microscopic observations and the function of the plasma membrane. This theory is called the fluid mosaic model. The model has evolved somewhat over time, but still best accounts for the structure and functions of the plasma membrane as we now understand them. The fluid mosaic model describes the structure of the plasma membrane as comprised of many different components (including phospholipids, cholesterol, proteins, and carbohydrates) that are able to flow and change position, while still maintaining the basic structure and integrity of the membrane. The plasma membrane is not a stiff, solid structure. It is quite fluid and both phospholipid molecules and embedded proteins are able to move side-to-side in the membrane. The fluidity of the plasma membrane is necessary for the activities of certain enzymes and transport molecules within the membrane. Despite the fluidity of the plasma membrane, it is rigid in that it does not allow the cell to change size easily (i.e. a cell cannot blow up like a balloon without damaging it).
Plasma membranes range from 5–10 nm (nanometers) thick. As a comparison, human red blood cells, visible via light microscopy, are approximately 8 μm (micrometers) thick, or approximately 1,000 times thicker than a plasma membrane.
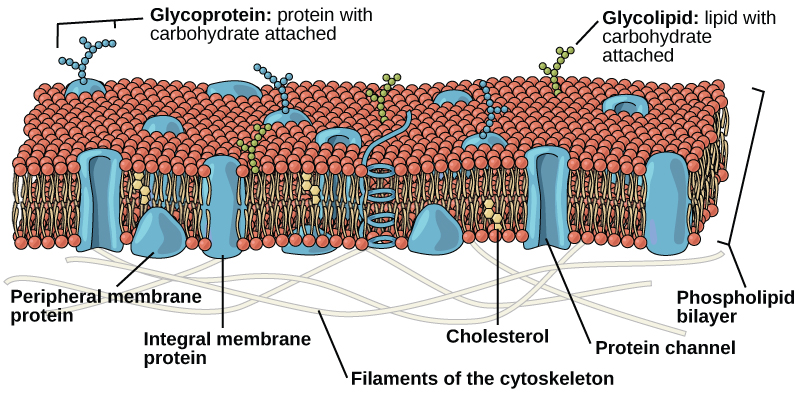
Components of the Plasma Membrane
The plasma membrane is made up primarily of a bilayer of phospholipids with embedded proteins, carbohydrates, glycolipids, and glycoproteins, and, in animal cells, cholesterol.
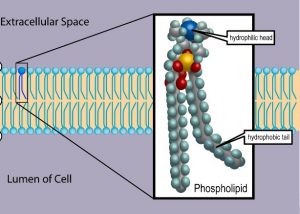
The main fabric of the membrane is composed of two layers of phospholipid molecules. Phospholipid molecules have a “head” that is hydrophilic (water-loving) and polar (Figure 2). Hydrophilic polar molecules “like” water or dissolve in water. Salt and sugar are examples of hydrophilic molecules. The hydrophilic polar heads of these molecules are in contact with the watery environment outside the cell as well as the cytoplasm (composed mostly of water) inside the cell. In contrast, the interior of the membrane, composed of the tails of phospholipids, is hydrophobic (water-fearing) or non-polar (Figure 2). Hydrophilic non-polar molecules do not “like” water or dissolve in water. Oil is an example of a hydrophobic molecule.
Proteins make up the second major chemical component of plasma membranes (Figure 1). Proteins are embedded in the plasma membrane and may go all the way through the membrane, or only be present on one side or the other. Proteins that go all the way through the membrane can serve as channels or pumps to move materials into or out of the cell. Other proteins are found on one side of the membrane or the other, but do not pass all the way through. Both types of proteins may serve as enzymes, as structural attachments for the fibers of the cytoskeleton, or as part of the cell’s recognition sites.
Carbohydrates are the third major component of plasma membranes (Figure 1). They are always found on the exterior surface of cells and are bound either to proteins (forming glycoproteins) or to lipids (forming glycolipids). These carbohydrate chains may consist of 2–60 monosaccharide units and may be either straight or branched. Along with peripheral proteins, carbohydrates form specialized sites on the cell surface that allow cells to recognize each other.
The amount of cholesterol in animal plasma membranes regulates the fluidity of the membrane and changes based on the temperature of the cell’s environment. In other words cholesterol acts as antifreeze in the cell membrane and is more abundant in animals that live in cold climates.
References
Unless otherwise noted, images on this page are licensed under CC-BY 4.0 by OpenStax.
Text adapted from: OpenStax, Concepts of Biology. OpenStax CNX. May 18, 2016 http://cnx.org/contents/b3c1e1d2-839c-42b0-a314-e119a8aafbdd@9.10

