Glucose Regulation and Utilization in the Body
On the last page, we traced the process of digesting the carbohydrates in a slice of pizza through the gastrointestinal tract, ending up with the absorption of monosaccharides across the cells of the small intestine and into the bloodstream. From there, they travel to the liver, where fructose and galactose are converted to glucose.
After any meal containing carbohydrates, you experience a rise in blood glucose that can serve as fuel for cells around the body. But during the periods between meals, including while you’re sleeping and exercising, your body needs fuel, too. To ensure that you have enough glucose in your blood at any given time, your body has a finely-tuned system to regulate your blood glucose concentration. This system allows you to store glucose when you have excess available (when your blood glucose is high) and to pull glucose out from your stores when needed (when your blood supply gets low).
Your body’s ability to maintain equilibrium or a steady state in your blood glucose concentration is called homeostasis. It’s a critical part of normal physiology, because if your blood glucose gets too low (called hypoglycemia, cellular function starts to fail, especially in the brain. If blood glucose gets too high (called hyperglycemia, it can cause damage to cells.
Hormones Involved in Blood Glucose Regulation
Central to maintaining blood glucose homeostasis are two hormones, insulin and glucagon, both produced by the pancreas and released into the bloodstream in response to changes in blood glucose.
- Insulin is made by the beta-cells of the pancreas and released when blood glucose is high. It causes cells around the body to take up glucose from the blood, resulting in lowering blood glucose concentrations.
- Glucagon is made by the alpha-cells of the pancreas and released when blood glucose is low. It causes glycogen in the liver to break down, releasing glucose into the blood, resulting in raising blood glucose concentrations. (Remember that glycogen is the storage form of glucose in animals.)
The image below depicts a mouse islet of Langerhans, a cluster of endocrine cells in the pancreas. The beta-cells of the islet produce insulin, and the alpha-cells produce glucagon.
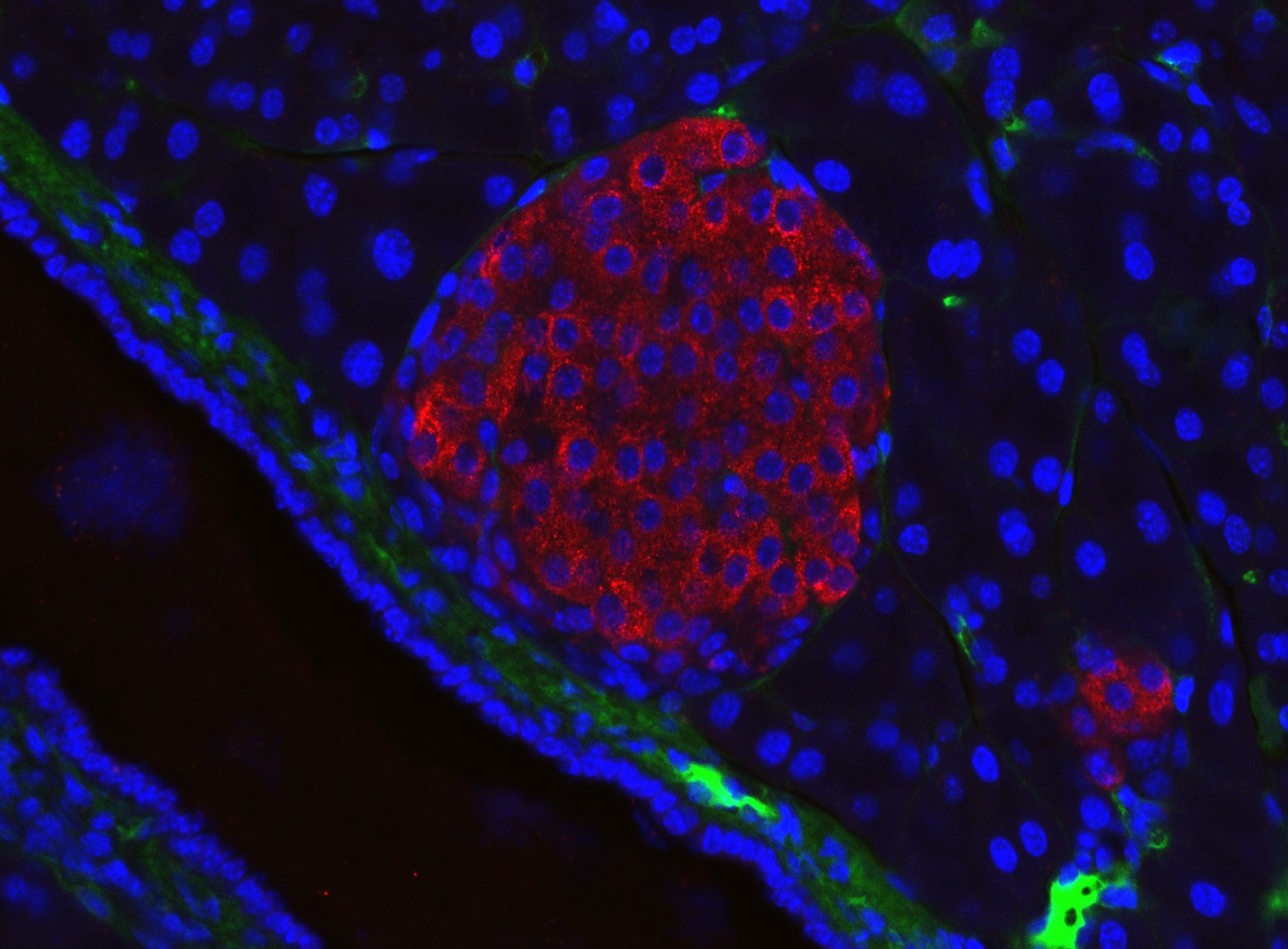
Figure 4.14. A mouse islet of Langerhans, visualized with immunofluorescent microscopy. In this image, cell nuclei are stained blue, insulin is stained red, and blood vessels are stained green. You can see that this islet is packed with insulin and sits right next to a blood vessel, so that it can secrete the two hormones, insulin and glucagon, into the blood. Glucagon is not stained in this image, but it’s there!
In the figure below, you can see blood glucose and insulin throughout a 24-hour period, including three meals. You can see that when glucose rises, it is followed immediately by a rise in insulin, and glucose soon drops again. The figure also shows the difference between consuming a sucrose-rich food and a starch-rich food. The sucrose-rich food results in a greater spike in both glucose and insulin. Because more insulin is required to handle that spike, it also causes a more precipitous decline in blood glucose. This is why eating a lot of sugar all at once may increase energy in the short-term, but soon after may make you feel like taking a nap!
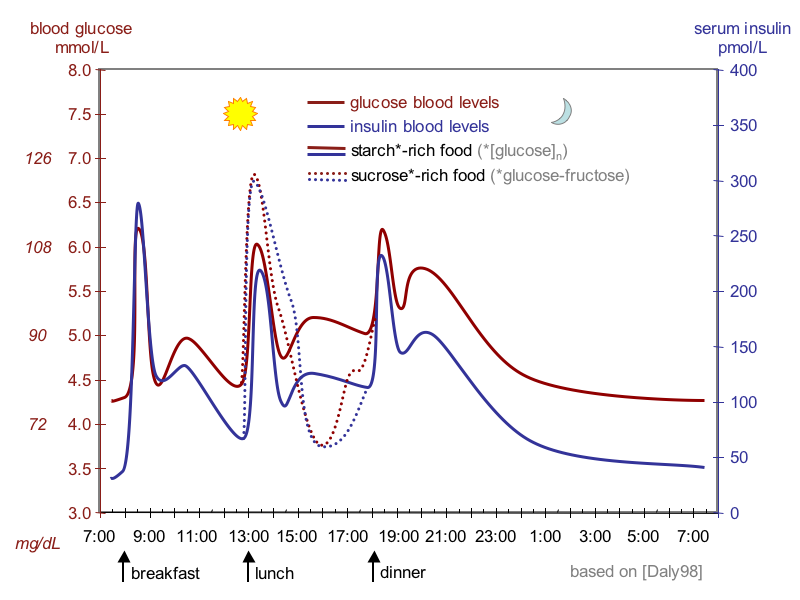
Figure 4.15. Typical pattern of blood glucose and insulin during a 24-hour period, showing peaks for each of 3 meals and highlighting the effects of consumption of sugar-rich foods.
Let’s look a little closer at how insulin works, illustrated in the figure below. Insulin is released by the pancreas into the bloodstream. Cells around the body have receptors for insulin on their cell membranes. Insulin fits into its receptors (labeled as step 1 in Figure 4.16), kind of like a key in a lock, and through a series of reactions (step 2), triggers glucose transporters to open on the surface of the cell (step 3). Now glucose can enter the cell, making it available for the cell to use and at the same time lowering the concentration of glucose in the blood.
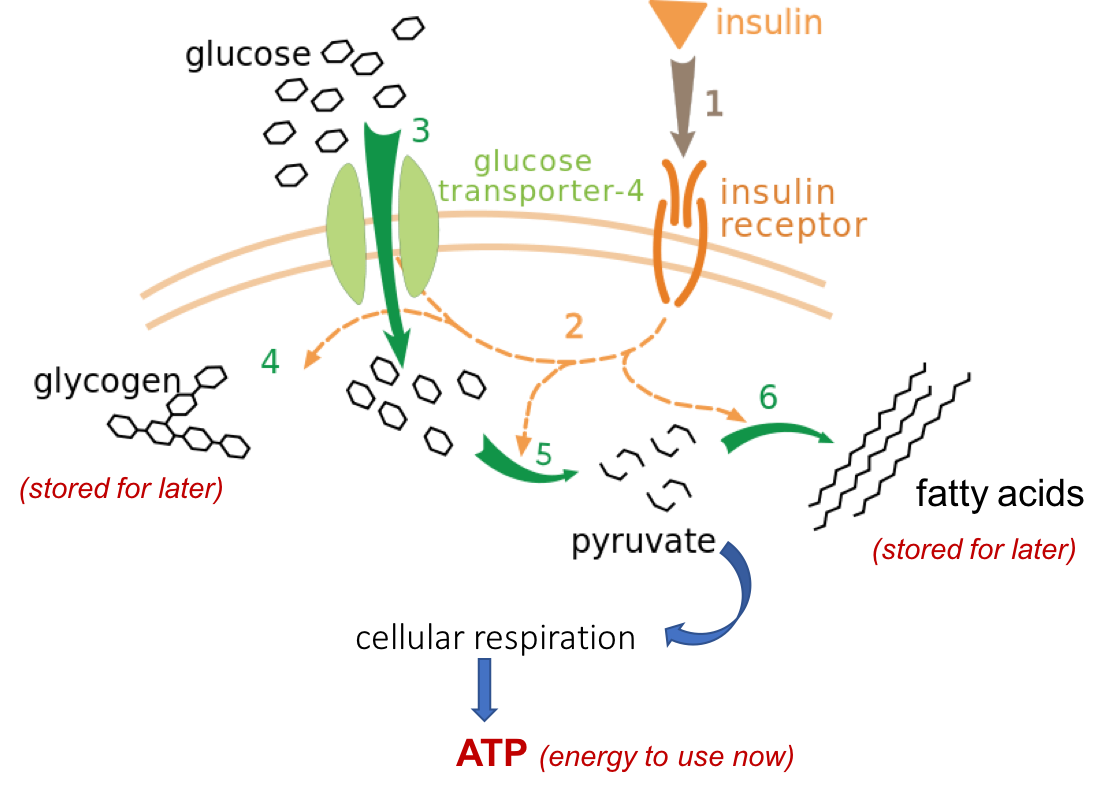
Figure 4.16. Insulin binds to its receptors on the cell membrane, triggering GLUT-4 glucose transporters to open on the membrane. This allows glucose to enter the cell, where it can be used in several ways.
The figure also shows several different ways glucose can be used once it enters the cell.
- If the cell needs energy right away, it can metabolize glucose through cellular respiration, producing ATP (step 5).
- If the cell doesn’t need energy right away, glucose can be converted to other forms for storage. If it’s a liver or muscle cell, it can be converted to glycogen (step 4). Alternatively, it can be converted to fat and stored in that form (step 6).
In addition to its role in glucose uptake into cells, insulin also stimulates glycogen and fat synthesis as described above. It also stimulates protein synthesis. You can think of its role as signaling to the body that there’s lots of energy around, and it’s time to use it and store it in other forms.
On the other hand, when blood glucose falls, several things happen to restore homeostasis.
- You receive messages from your brain and nervous system that you should eat.
- Glucagon is released from the pancreas into the bloodstream. In liver cells, it stimulates the breakdown of glycogen, releasing glucose into the blood.
- In addition, glucagon stimulates a process called gluconeogenesis, in which new glucose is made from amino acids (building blocks of protein) in the liver and kidneys, also contributing to raising blood glucose.
How Glucose Provides Energy
Now let’s zoom in on how exactly glucose provides energy to the cell. We can trace this process in the figure below.
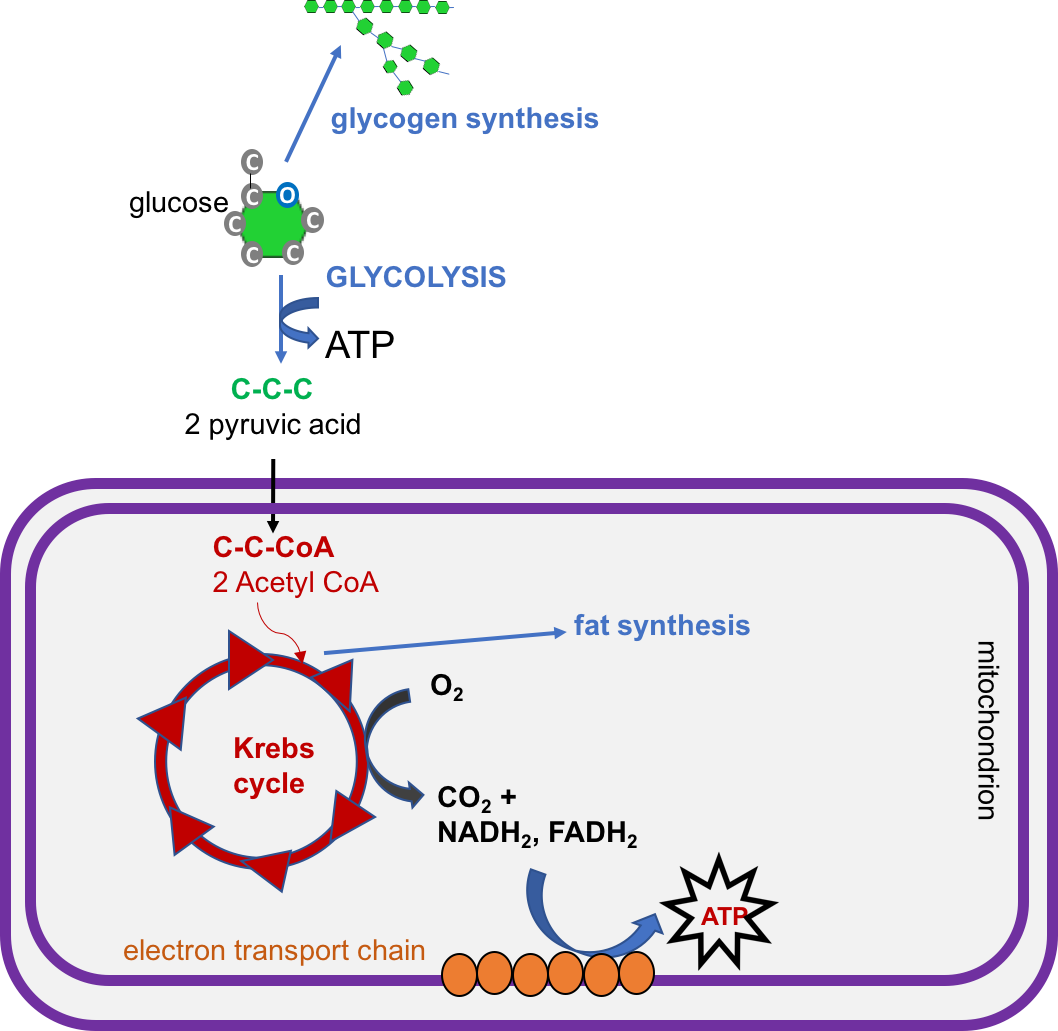
Figure 4.17. Overview of glucose metabolism in the fed state, when there is adequate glucose available. Glucose can be used to generate ATP for energy, or it can be stored in the form of glycogen or converted to fat for storage in adipose tissue.
- Glucose, a 6-carbon molecule, is broken down to two 3-carbon molecules called pyruvate through a process called glycolysis.
- Pyruvate enters a mitochondrion of the cell, where it is converted to a molecule called acetyl CoA.
- Acetyl CoA goes through a series of reactions called the Krebs cycle. This cycle requires oxygen and produces carbon dioxide. It also produces several important high energy electron carriers called NADH2 and FADH2.
- These high energy electron carriers go through the electron transport chain to produce ATP—energy for the cell!
- Note that the figure also shows that glucose can be used to synthesize glycogen or fat, if the cell already has enough energy.
What Happens When There Isn’t Enough Glucose?
We’ve already talked about what happens when blood glucose falls: glucagon is released, and that stimulates the breakdown of glycogen as well as the process of gluconeogenesis from amino acids. These are important mechanisms for maintaining blood glucose levels to fuel the brain when carbohydrate is limited. Hypoglycemia (low blood glucose) can cause you to feel confused, shaky, and irritable, because your brain doesn’t have enough glucose. If it persists, it can cause seizures and eventually coma, so it’s good we have these normal mechanisms to maintain blood glucose homeostasis!
What happens if your carbohydrate supply is limited for a long time? This might happen if a person is fasting, starving, or consuming a very low carbohydrate diet. In this case, your glycogen supplies will become depleted within about 24 hours. How will you get enough glucose (especially for the brain) and energy? You’ll have to use the other two macronutrients in the following ways:
- Protein: You’ll continue to use some amino acids to make glucose through gluconeogenesis and others as a source of energy through acetyl CoA. However, if a person is starving, they also won’t have extra dietary protein. Therefore, they start breaking down body proteins, which will cause muscle wasting.
- Fat: You can break down fat as a source of energy, but you can’t use it to make glucose. Fatty acids can be broken down to acetyl CoA in the liver, but acetyl CoA can’t be converted to pyruvate and go through gluconeogenesis. It can go through the Krebs cycle to produce ATP, but if carbohydrate is limited, the Krebs cycle gets overwhelmed. In this case, acetyl CoA is converted to compounds called ketones or ketone bodies. These can then be exported to other cells in the body, especially brain and muscle cells.
These pathways are shown in the figure below:
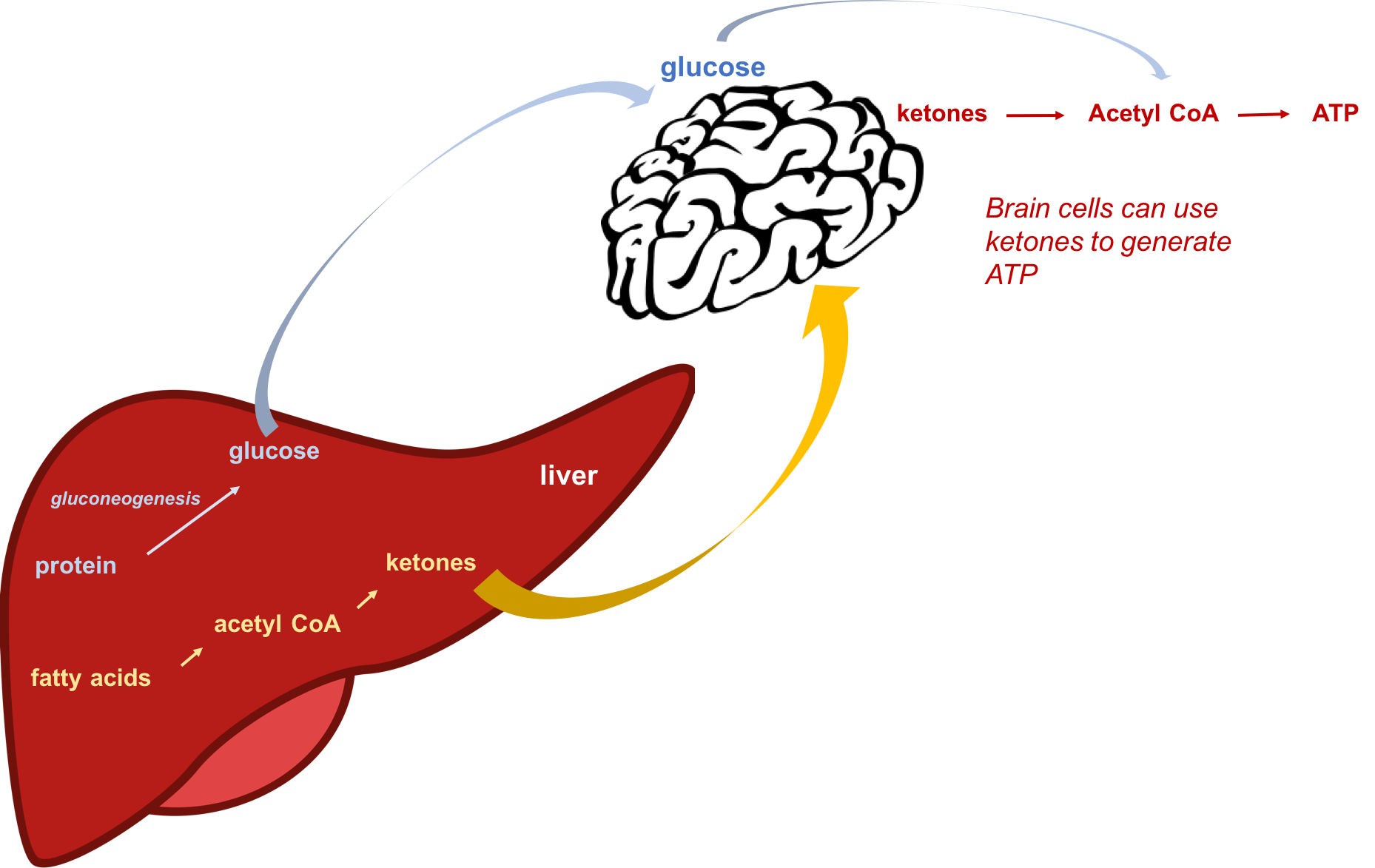
Figure 4.18. During starvation or when consuming a low-carbohydrate diet, protein (amino acids) can be used to make glucose by gluconeogenesis, and fats can be used to make ketones in the liver. The brain can adapt to using ketones as an energy source in order to conserve protein and prevent muscle wasting.
Ketone production is important, because ketones can be used by tissues of the body as a source of energy during starvation or a low carbohydrate diet. Even the brain can adapt to using ketones as a source of fuel after about three days of starvation or very low-carbohydrate diet. This also helps to preserve the protein in the muscle.
Ketones can be excreted in urine, but if ketone production is very high, they begin to accumulate in the blood, a condition called ketosis. Symptoms of ketosis include sweet-smelling breath, dry mouth, and reduced appetite. People consuming a very low carbohydrate diet may be in ketosis, and in fact, this is a goal of the currently popular ketogenic diet. (Ketones are acidic, so severe ketosis can cause the blood to become too acidic, a condition called ketoacidosis. This mainly happens with uncontrolled diabetes.)
Is following a ketogenic diet an effective way to lose weight? It can be, but the same can be said of any diet that severely restricts the types of foods that you’re allowed to eat. Following a ketogenic diet means eating a high fat diet with very little carbohydrate and moderate protein. This means eating lots of meat, fish, eggs, cheese, butter, oils, and low carbohydrate vegetables, and eliminating grain products, beans, and even fruit. With so many fewer choices, you’re likely to spend more time planning meals and less time mindlessly snacking. Being in ketosis also seems to reduce appetite, and it causes you to lose a lot of water weight initially. However, studies show that being in ketosis doesn’t seem to increase fat-burning or metabolic rate. There are also concerns that the high levels of saturated fat in most ketogenic diets could increase risk of heart disease in the long term. Finally, it’s a very difficult diet to maintain for most people, and reverting back to your previous dietary patterns usually means the weight will come back. The ketogenic diet is also very similar to the Atkins diet that was all the rage in the 1990’s, and we tend to be skeptical of such fad diets, preferring to focus instead on balance, moderation, and enjoyment of a wide variety of foods.
VIDEO: “Glucose Regulation and Utilization in the Body,” by Alice Callahan, YouTube (October 1, 2018), 13:11 minutes.
Diabetes
Diabetes is a chronic disease in which your normal system of regulating blood glucose doesn’t work. There are three main types of diabetes: type 1, type 2, and gestational diabetes.
Type 1 Diabetes:
This is an autoimmune disease in which the beta-cells of the pancreas are destroyed by your own immune system. Without the beta-cells, you can’t make enough insulin, so in type 1 diabetes, you simply don’t have enough insulin to regulate your blood glucose levels. Remember how we said insulin is like the key that lets glucose into the body’s cells? In type 1 diabetes, you’re missing the key, so glucose stays in the blood and can’t get into cells.
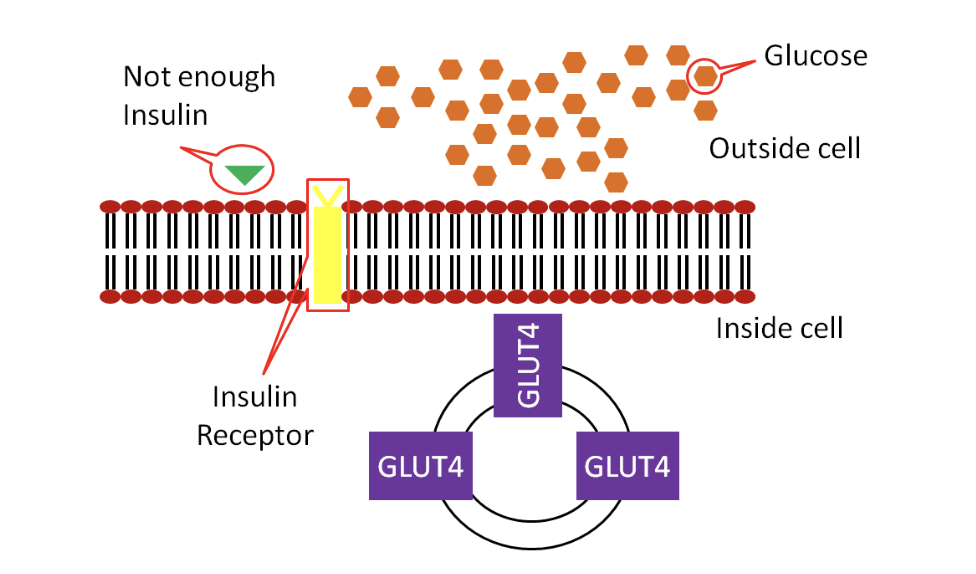
Figure 4.19. In type 1 diabetes, the pancreas does not make enough insulin, so glucose transporters (GLUT-4) do not open on the cell membrane, and glucose is stuck outside the cell.
Common symptoms include weight loss and fatigue, because the body’s cells are starved of glucose. Excess glucose from the blood is also excreted in the urine, increasing urination and thirst.
Once diagnosed, type 1 diabetics have to take insulin in order to regulate their blood glucose. Traditionally, this has required insulin injections timed with meals. New devices like continuous glucose monitors and automatic insulin pumps can track glucose levels and provide the right amount of insulin, making managing type 1 diabetes a little easier. Figuring out the right amount of insulin is important, because chronically elevated blood glucose levels can cause damage to tissues around the body. However, too much insulin will cause hypoglycemia, which can be very dangerous.
Type 1 diabetes is most commonly diagnosed in childhood, but it has been known to develop at any age. It’s much less common than type 2 diabetes, accounting for 5-10% of cases of diabetes.
VIDEO: “What is Type 1 Diabetes?” by Diabetes UK, YouTube (April 10, 2018), 2:27 minutes.
Type 2 Diabetes:
Development of type 2 diabetes begins with a condition called insulin resistance. At least initially, the pancreas is producing enough insulin, but the body’s cells don’t respond appropriately. It’s as if you still have the insulin key but can’t find the keyhole to unlock the doors and let the glucose in.
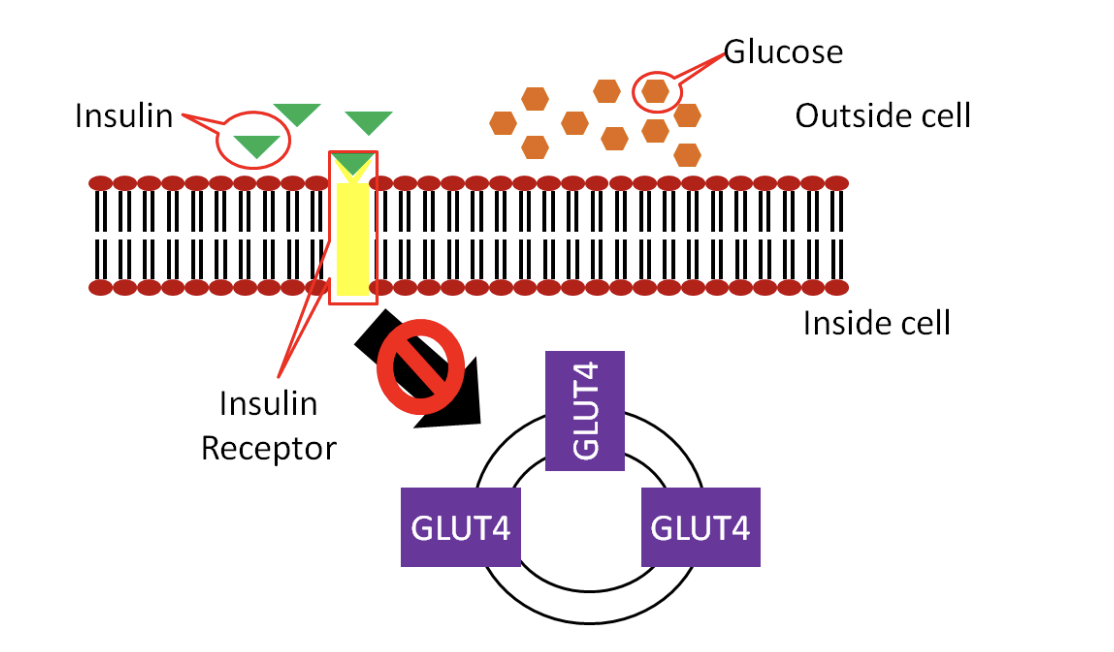
Figure 4.20. In type 2 diabetes, the cell does not respond appropriately to insulin, so glucose is stuck outside the cell.
The result is the same: high blood glucose. At this point, you may be diagnosed with a condition called prediabetes. The pancreas tries to compensate by making more insulin, but over time, it becomes exhausted and eventually produces less insulin, leading to full-blown type 2 diabetes. According to the CDC, 100 million Americans are living with diabetes (30.3 million) or prediabetes (84.1 million).
Although people of all shapes and sizes can get type 2 diabetes, it is strongly associated with abdominal obesity. In the past, it was mainly diagnosed in older adults, but it is becoming more and more common in children and adolescents as well, as obesity has increased in all age groups. In the maps below, you can see that as obesity has increased in states around the country, so has diabetes.
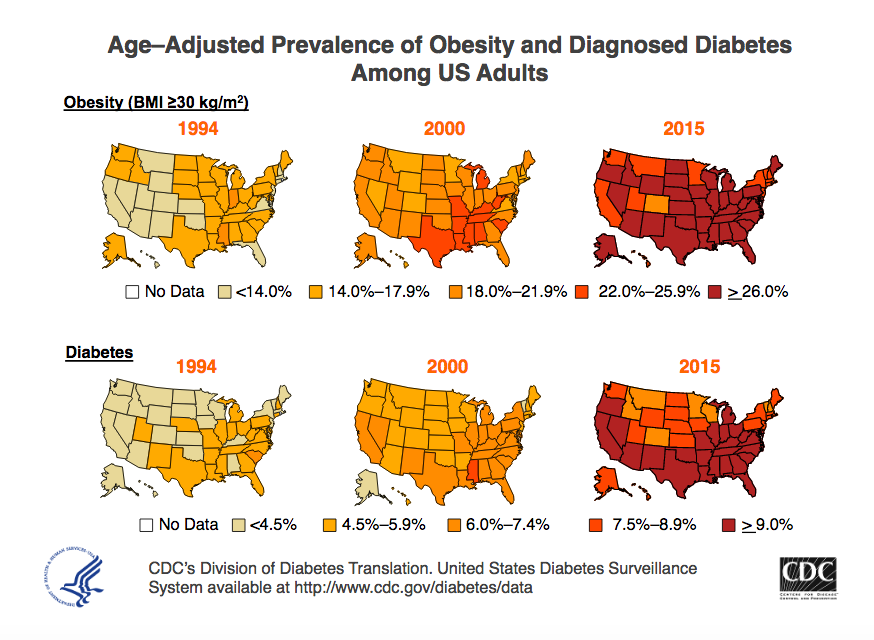
Figure 4.21. Data from the CDC show the increasingly prevalence of both obesity and type 2 diabetes between 1994 and 2015.
The complications of type 2 diabetes result from long-term exposure to high blood glucose, or hyperglycemia. This causes damage to the heart, blood vessels, kidneys, eyes, and nerves, increasing the risk of heart disease and stroke, kidney failure, blindness, and nerve dysfunction. People with uncontrolled Type 2 diabetes can also end up with foot infections and ulcers because of impaired nerve function and wound healing. If left untreated, this results in amputation.
VIDEO: “What is Type 2 Diabetes?” by Diabetes UK, YouTube (April 10, 2018), 2:36 minutes. This video reviews the causes, complications, and treatments for type 2 diabetes.
VIDEO: “Obesity and Type 2 Diabetes (HBO: The Weight of the Nation)”, by HBO Docs, YouTube (May 14, 2012), 15:20 minutes.
Gestational diabetes:
Gestational diabetes is diabetes that develops during pregnancy in women that did not previously have diabetes. It affects approximately 6 percent of pregnancies in the U.S. It can cause pregnancy complications, mostly associated with excess fetal growth because of high blood glucose. Although it usually goes away once the baby is born, women who have gestational diabetes are more likely to develop type 2 diabetes later in life, so it is a warning sign for them.6
VIDEO: “Types of Diabetes” by KhanAcadamyMedicine, YouTube (May 14, 2015), 5:57 minutes. This video does a nice job of explaining the causes of the different types of diabetes.
Diabetes Management:
All of the following have been shown to help manage diabetes and reduce complications. Diabetes management, as well as prevention (particularly if you’ve been diagnosed with prediabetes), starts with lifestyle choices.
- Exercise helps to improve your body’s insulin response and can also help maintain a healthy weight.
- Eating well with diabetes doesn’t require a special diet but instead regular, balanced meals following the Dietary Guidelines. It isn’t necessary to eliminate carbohydrates or eat a low-carbohydrate diet, but emphasizing whole food sources of carbohydrate helps with blood glucose regulation.
- Managing stress levels and getting enough sleep can also help with blood glucose regulation.
- Medications may be needed. Insulin is needed for type 1 diabetes and may be needed for more advanced or severe cases of type 2 or gestational diabetes. Other medications can also help. If lifestyle choices aren’t enough to manage diabetes, it is important to use medications appropriately to help reduce the complications of chronic high blood glucose.
Self-Check:
References:
- Salway, J. G. (2004). Metabolism at a Glance (3rd ed.). Malden, Mass.: Blackwell Publishing.
- Smolin, L., & Grosvenor, M. (2016). Nutrition Science and Applications. Danvers, Mass.: John Wiley & Sons, Ltd.
- 1Gibson, A. A., Seimon, R. V., Lee, C. M. Y., Ayre, J., Franklin, J., Markovic, T. P., … Sainsbury, A. (2015). Do ketogenic diets really suppress appetite? A systematic review and meta-analysis. Obesity Reviews: An Official Journal of the International Association for the Study of Obesity, 16(1), 64–76. https://doi.org/10.1111/obr.12230
- 2Hall, K. D., Chen, K. Y., Guo, J., Lam, Y. Y., Leibel, R. L., Mayer, L. E., … Ravussin, E. (2016). Energy expenditure and body composition changes after an isocaloric ketogenic diet in overweight and obese men. The American Journal of Clinical Nutrition, 104(2), 324–333. https://doi.org/10.3945/ajcn.116.133561
- 3Abbasi, J. (2018). Interest in the Ketogenic Diet Grows for Weight Loss and Type 2 Diabetes. JAMA, 319(3), 215–217. https://doi.org/10.1001/jama.2017.20639
- 4Belluz, J. (2018, February 21). The keto diet, explained. Retrieved November 15, 2019, from Vox website: https://www.vox.com/science-and-health/2018/2/21/16965122/keto-diet-reset
- 5Centers for Disease Control and Prevention. (2019, June 11). Diabetes Basics. Retrieved November 15, 2019, from https://www.cdc.gov/diabetes/basics/index.html
- 6Deputy, N. P., Kim, S. Y., Conrey, E. J. & Bullard, K. M. Prevalence and Changes in Preexisting Diabetes and Gestational Diabetes Among Women Who Had a Live Birth — United States, 2012–2016. MMWR Morb Mortal Wkly Rep 67, (2018). https://www.cdc.gov/mmwr/volumes/67/wr/mm6743a2.htm
Image Credits:
- Figure 4.14. “Mouse pancreatic Islet” by Jakob Suckale is licensed under CC BY-SA 3.0
- Figure 4.15. “Glucose/insulin patterns in 24-hours” by Jakob Suckale and Michele Solimena, is licensed under CC BY 3.0
- Figure 4.16. “Insulin glucose metabolism” by Alice Callahan is licensed under CC BY-NC-SA 4.0 / A derivative from the original work
- Figure 4.17. “Glucose metabolism” by Alice Callahan is licensed under CC BY-NC-SA 4.0, with “ATP star” by Anastasia Latysheva is in the Public Domain
- Figure 4.18. “Gluconeogenesis and ketogenesis” by Alice Callahan is licensed under CC BY-NC-SA 4.0, with “brain” by monstara and “liver” by maritacovarrubias, both in the public domain
- Figure 4.19 and 4.20. “Type 1 diabetes” and “Type 2 diabetes” by Brian Lindshield, “Kansas State University Human Nutrition (FNDH 400) Flexbook” is licensed under CC BY-NC-SA 4.0
- Figure 4.21. “Prevalence of Obesity” by the CDC is in the Public Domain
The body’s ability to maintain equilibrium or a steady state, such as maintaining blood glucose concentration.
Low blood sugar.
High blood sugar.
A hormone made by the pancreas and released when blood glucose is high; it results in lowering blood glucose concentrations.
A hormone made by the pancreas and released when blood glucose is low; it causes glycogen in the liver to break down, results in raising blood glucose concentrations.
The synthesis of new glucose molecules from amino acids.
A 3-carbon molecule that is the end product of glycolysis.
A cycle that occurs in the cytoplasm of the cell and plays a central role in the production of energy through anaerobic metabolism.
An important molecule for many metabolic pathways, including the Krebs Cycle in aerobic metabolism.
A cycle that occurs in the mitochondria of cells and plays a central role in the production of energy through aerobic metabolism.
Compounds that are made when fatty acid breakdown is high and glucose is limited.
Another name for ketones.
The accumulation of ketones in the blood.
A condition which results from too many ketones accumulating in the blood, resulting in the blood being too acidic.
A chronic disease in which the body isn’t able to regulate blood glucose; includes type 1, type 2, and gestational diabetes.
An autoimmune disease in which the cells of the pancreas that create insulin are destroyed.
The most common type of diabetes, occurring when cells stop responding to insulin; strongly associated with abdominal obesity.
A condition where cells stop responding to insulin.
A condition that involves insulin resistance, but not full-blown type 2 diabetes.
A type of diabetes that develops during pregnancy in women who did not previously have diabetes.

