19 The Scientific Process
Science as a Cycle
Using the scientific method as the hub of a cyclic process has led to the massive rate of scientific and technological advancement we have seen over the last two centuries. Science can be thought of as a continuous process guided by with the scientific method as discussed in the following video:
Reinforcement Exercise
Modern science is done according to a complex process of checks and balances, such as replication and peer-review. This complexity emerged to help ensure the integrity of scientific results, but the process remains rooted in the basic scientific method. You can apply the basic scientific method every day, just as Jolene did, in order to ensure that you make informed decisions that aren’t overly biased by inaccurate data, false logic, or your own preconceptions.
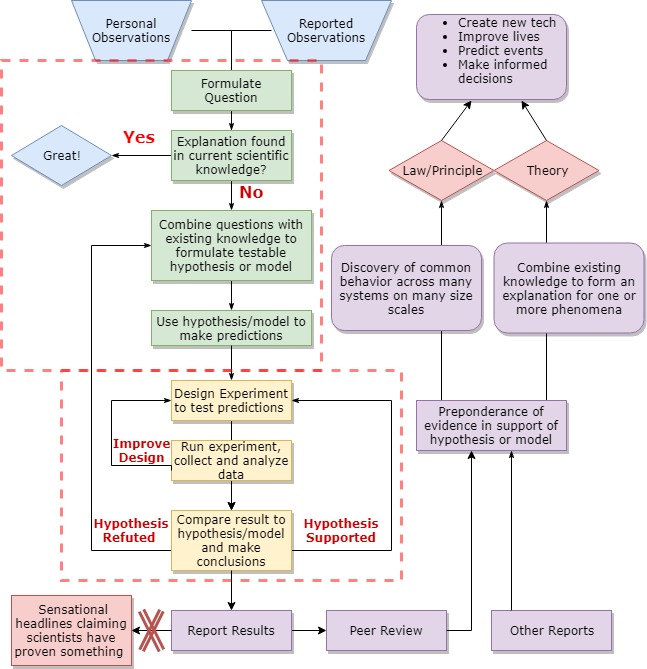
The previous diagram illustrates the complex scientific process, but also highlights the basic scientific method that Jolene used in the previous example, on which the whole process is built. After observation, the basic scientific method follows the green and yellow boxes within the dotted line in the diagram below. Generally speaking, the green boxes comprise theoretical science and the yellow boxes comprise experimental science. These days most scientists participate in some or all parts of both categories and collaborate with other scientists to complete the process.
The uncertainty associated with all measurements means that science cannot prove anything, despite what the media often claims. Instead, the scientific process produces reviewed and reproduced conclusions that account for uncertainty. (We will learn how scientists recognize and deal with uncertainty in the next chapter). Scientific conclusions provide evidence for or against hypotheses.
Laws, Principles and Theories
Laws
When a certain behavior is repeatedly observed across many systems of many sizes and time periods, then the behavior becomes a law. A law is not an explanation of the observed behavior. For example, the 1st Law of Thermodynamics states that the when a system does work and/or loses heat, the internal energy of the system must drop by an amount equal to the work done plus the heat lost.
Principles
Principles summarize rules created based on collections of laws and followed by scientists when formulating hypotheses, designing experiments, analyzing results. For example, the principle of conservation of energy states that energy cannot be created or destroyed, only transferred. The 1st Law of Thermodynamics supports the principle of conservation of energy.
Theories
When a preponderance of evidence supports a particular explanation for observed occurrences (phenomena), then the explanation becomes a theory. Laws, principles, and theories are what the general public and media often refer to as scientific facts, but we don’t need to introduce another definition so we won’t use fact here. We will combine and apply a variety of laws, principles, and theories to understand how the body functions.
Reinforcement Exercises
prejudice in favor of or against one thing, person, or group compared with another, usually in a way considered to be unfair or inaccurate.
a method of procedure that has characterized natural science since the 17th century, consisting in systematic observation, measurement, and experiment, and the formulation, testing, and modification of hypotheses
a proposed explanation made on the basis evidence that can be supported or refuted by the result of experimentation
a statement, usually in the form of a mathematical equation, that summarizes, but not explains, the results of repeated experiments or observations that describe some aspect of the natural, usually within a certain range of application.
Any change in the internal energy of a system must in the process of exchanging heat, doing work, or both.
an explanation of an aspect of the natural world that can be repeatedly tested and verified in accordance with the scientific method using accepted standard protocols

